Author: Roy Sebastian MD, DPT, MBA*1, Alan D. Proia MD 2, Sweta Sengupta MD 3
Author Affiliation:
1 Department of Anesthesiology (Pain medicine), University of Maryland Medical Center, Baltimore, MD.
2 Department of Pathology, Duke University Medical Center, Durham, NC, USA, and Department of Pathology, Campbell University Jerry M. Wallace School of Osteopathic Medicine, Lillington, NC.
3 Department of Neurology, Duke University Medical Center, Durham, NC.
Competing Interests: The author/s declare no competing interests.
Issue: 16.02
DOI: 10.30756/ahmj.2025.16.02
Received: Sept 11, 2025
Accepted: Nov 12, 2025
Published: Dec 1, 2025
Recommended Citation: Sebastian R, Proia AD, Sengupta S. Giant Cell Arteritis With Unremarkable Inflammatory Markers And Imaging In A Patient With Post-Concussion Syndrome: Highlighting The Role Of Early Biopsy And Steroid Treatment. Ann Head Med. 2025;16:02. DOI: 10.30756/ahmj.2025.16.02
Giant cell arteritis (GCA) is an immune-mediated, systemic granulomatous vasculitis that targets large and medium-sized arteries. Vision loss and stroke are potential permanent consequences of GCA if immediate treatment is not provided. Diagnosing GCA in a patient presenting with headache in the context of post-concussion syndrome poses a challenge because of overlapping clinical features and the non-specific nature of diagnostic studies. Following a motor vehicle accident (MVA) in 2021, a man developed a new persistent headache over the right temple with photophobia and phonophobia. Fourteen months later, his localized headache worsened after another head injury. Neither the initial ultrasound nor the blood tests for inflammatory markers were positive for GCA. However, the patient was diagnosed with GCA after undergoing a temporal artery biopsy in August 2023. Symptom improvement occurred for the patient following the start of steroid treatment. Post-concussion headaches can obscure timely diagnosis and delay treatment of GCA.
Introduction
Giant cell arteritis is a common vasculitis that primarily affects individuals over 50 years of age and is characterized by inflammation of large and medium-sized arteries. Approximately, 22% of GCA patients do not experience headaches.1 In those who do, International Classification of Headache Disorders version 3 (ICHD-3) labels this headache type as “Headache attributed to giant cell arteritis”.2 ICHD-3 also mentions the term “Persistent headache attributed to traumatic injury to the head” and “Persistent headache attributed to whiplash” and does not specifically use “post-concussion headaches” to define headaches after a concussion event. Treating patients with overlapping types requires comprehensive, multidisciplinary care and close monitoring to adjust treatment plans as symptoms evolve.
Case Report
A man in his 60s with major depressive disorder, hyperlipidemia, and hypertension presented after an MVA in September 2021. After the accident, he developed new, persistent right temple headaches with associated photophobia and phonophobia, vertigo, anxiety, nightmares, and irritability. He had been taking ibuprofen 600-800 mg at least once daily since then which provided partial relief with intensity. His symptoms worsened after a moderate second head injury in November 2022, which exacerbated his headaches, nausea, and dizziness. The headaches evolved to involve the entire head but remained more pronounced in the right temporal area. He was diagnosed with post-concussive syndrome. He also struggled with balance issues, requiring assistive devices such as a cane or walker, and underwent physical therapy. Despite medical management with gabapentin and verapamil along with vestibular physical therapy, his condition only slightly improved, and his holocephalic headache remained constant, often intensifying by the evening. Additionally, we started the patient on venlafaxine for headache management and anxiety. In early 2023, he was diagnosed with moderate obstructive sleep apnea (OSA) and subsequently began using a continuous positive airway pressure (CPAP) machine. While the CPAP improved his sleep, he continued to experience daytime sleepiness, fatigue, headache, dizziness, cognitive difficulties, and photophobia. Due to intermittent right temple throbbing pain, blurred vision, and jaw fatigue when chewing, preliminary work-up for giant cell arteritis was completed. Initial GCA ultrasound and blood tests [Erythrocyte sedimentation rate (ESR); 19, C-reactive protein (CRP): 0.8, platelets 215×109/L, and white blood cell 5.6 x109/L, Aspartate Aminotransferase (AST):27, Alanine Aminotransferase (ALT): 40] were unrevealing, except for mild ALT elevation.
In July 2023, he reported intensification of the pain over the left temple in addition to the right. Repeated ESR, CRP, and temporal artery ultrasound tests were unremarkable. Follow-up physical examination revealed a firmer, tender right temporal artery. Patient was prescribed Prednisone 40 mg for four days due to the suspicion for GCA and a temporal artery biopsy was ordered. He underwent a bilateral temporal artery biopsy which is considered the gold standard for diagnosis of GCA, five days after completing the prednisone 40 mg. The superficial temporal artery biopsy demonstrated healing arteritis and he was started on prednisone 60 mg daily by the Ophthalmologist. With the high doses of prednisone, the temporal headaches became mild, but he continued to experience daily holocephalic headaches, blurred vision, photophobia, and phonophobia. Multiple medications were used for both prevention and acute treatment of his post-concussive headaches with migrainous features, including verapamil, propranolol, sumatriptan, rizatriptan, naratriptan, venlafaxine, and meclizine with inadequate response.
In August 2023, he started Eptinezumab infusion, a calcitonin gene-related peptide antagonist, due to inadequate response to current medical management for chronic headaches with migraine features. His prednisone dosage was gradually tapered, but he continued to experience mild persistent right temporal pain. By June 2024, his headaches had significantly reduced with Eptinezumab. By early 2025, his right temporal headaches became less intense, and his inflammatory markers remained normal, allowing for the discontinuation of prednisone. His condition stabilized, reflecting significant improvements in both GCA and headache management despite ongoing challenges. He continued his rehabilitation and therapy, with his condition remaining stable as of early 2025.
Investigations
The comprehensive series of investigations provided essential insights into the patient’s condition and played a key role in guiding treatment decisions. The magnetic resonance imaging (MRI) of the brain with and without contrast (March 2022) revealed no acute intracranial abnormalities, such as infarction or hemorrhage, but highlighted mild chronic microvascular ischemic changes, a finding that is often associated with aging and cardiovascular risk factors.
In September 2022, he was diagnosed with moderate OSA with a respiratory event index (REI) of 17/hr, which may have contributed to his fatigue and cognitive issues. Vestibular testing in December 2022 revealed right vestibular hypofunction, which may explain his complaints of dizziness and balance issues and supported the need for vestibular rehabilitation.
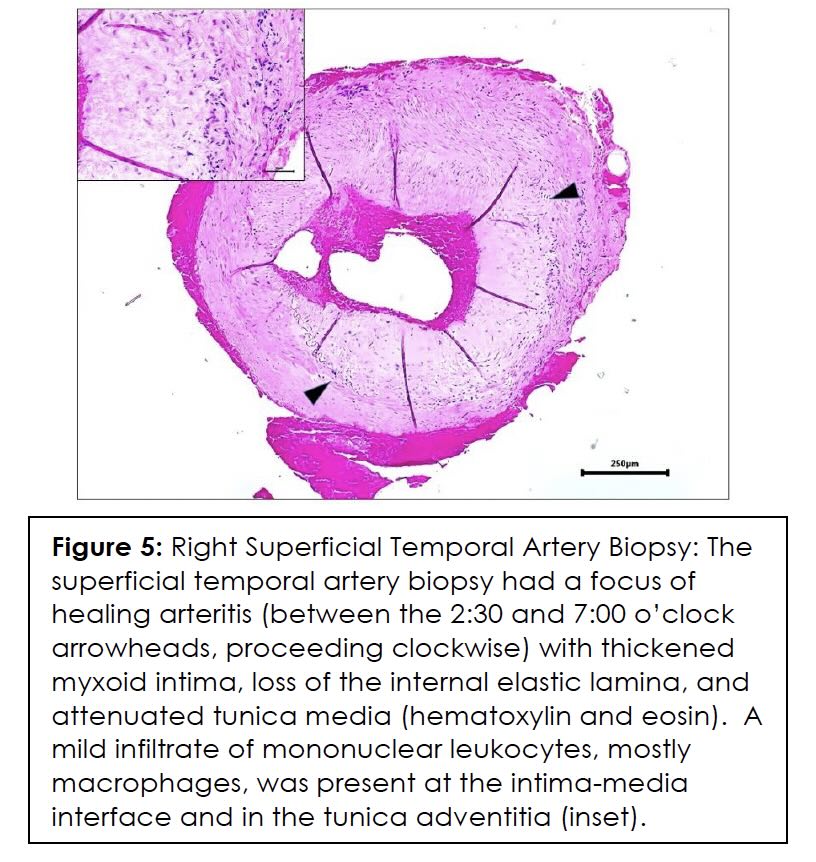
ESR, CRP, and color Doppler ultrasound (CDUS) of his temporal artery in July 2023 were negative, showing no GCA in the superficial temporal arteries. CDUS of bilateral temporal arteries demonstrated normal flow characteristics (Figures 1,2,3,4). Physical examination and temporal predominance of headache prompted obtaining the temporal artery biopsy in August 2023, which revealed healing GCA (Figure 5). The presence of myxoid change in the asymmetrically thickened intima and a residual infiltrate of mostly macrophages at the tunica intima-tunica media interface where the internal elastic lamina was absent allowed distinction of healing arteritis from age-related histopathological change, which may resemble healed giant cell arteritis. 3
Later, imaging studies were obtained due to persistence of headache despite medical management and high-dose prednisone. The MRI brain and magnetic resonance angiogram (MRA) of the head with and without contrast (August 2023) showed no new intracranial abnormalities and confirmed the presence of moderate temporomandibular joint osteoarthrosis, which may have contributed to some of the patient’s jaw pain. MRI of the brain without contrast (May 2024) demonstrated no new findings. Together, these investigations formed a comprehensive picture of the patient’s complex medical condition and directed ongoing treatment.
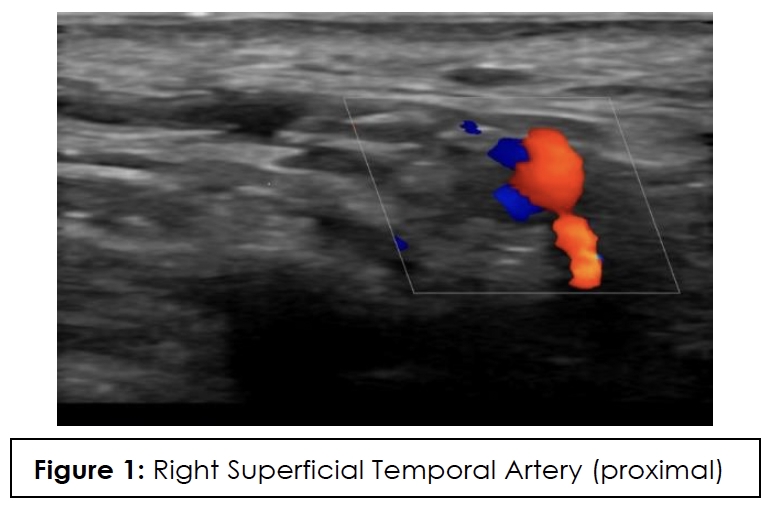
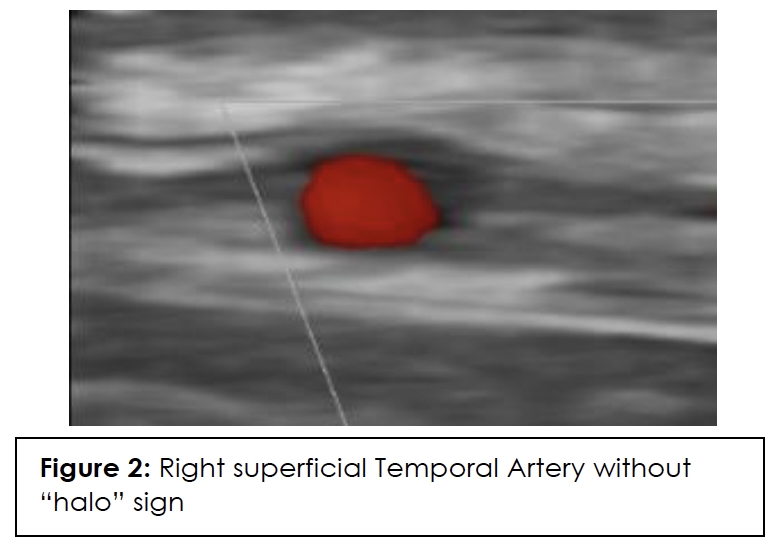
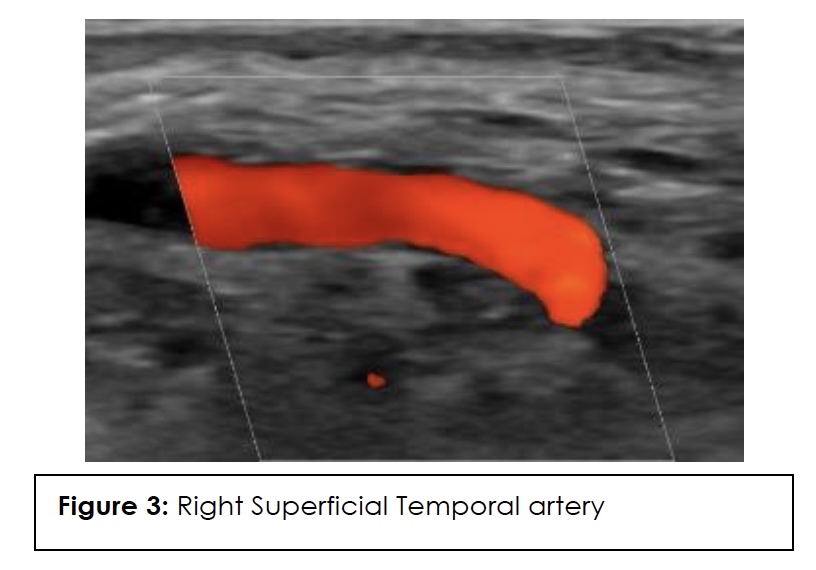
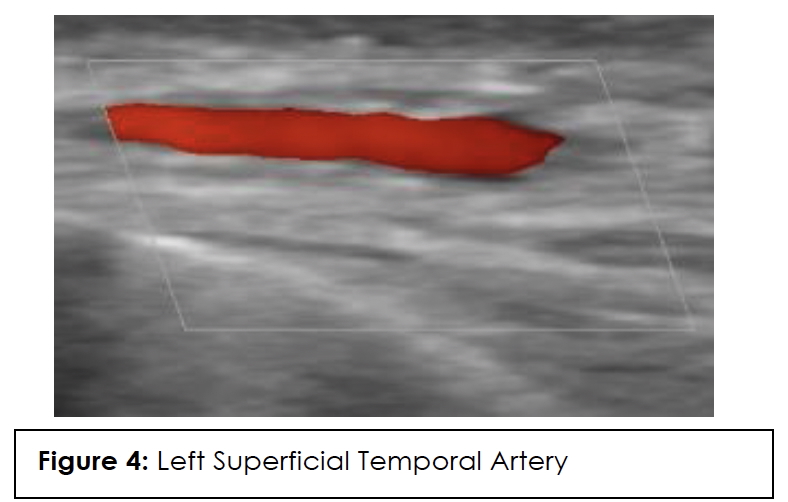
Differential Diagnoses
Multiple conditions were considered in the patient’s differential diagnosis, including migraine chronification, cervicogenic headache, temporal mandibular joint dysfunction, scalp allodynia, untreated sleep apnea, lesser occipital neuralgia, and tension-type headache. The patient’s initial headache was unilateral but later became bilateral, involving both temples.
A study 4 using data from the American Registry for Migraine Research found that patients with a history of mild traumatic brain injury (mTBI) had a significantly higher prevalence of chronic migraine compared to those without TBI. Specifically, 74.3% of patients with mild TBI (307 of 413) reported chronic migraines, versus 65.8% of non-TBI patients (422 out of 641), a difference that was statistically significant (p = 0.004). Given that the patient has no prior history of migraine and that all headaches began following the MVA, it is not reasonable to attribute the headaches to migraine chronification.
The patient’s MRI showed minimal bilateral TMJ degenerative change. TMJ dysfunction and excessive chewing is known to present with temporal pain. 5 However, jaw claudication can also present in GCA related to impaired blood flow to masticatory muscles. A lack of electrical component in the patient’s pain description helped to distinguish it from lesser occipital neuralgia, which is a neuropathic pain that radiates around the ear from posterior scalp.
Our patient reported right temporal cutaneous allodynia. Scalp allodynia-pain resulting from normally non-painful stimuli- can occur in 60-80% 6 of those with migraine, 53.8% of those with TBI, and in cases of GCA. 7-9 This phenomenon is thought to be due to neurons propagating pain signals in the absence of external stimuli (central sensitization). The absence of typical morning headaches and presence of continuous headache pattern reduces likelihood of headache attributed to OSA.10 Tension-type headache is typically bilateral pain in nature; however, our patient reported bilateral temporal pain, which would contribute to difficulty with distinguishing our patient’s headache type from tension-type headache (TTH).11 In addition, our patient had migraine features to his headache type, which would not align with TTH as a diagnosis. Dash et al. 2023, described a case of a patient with giant cell arteritis (GCA) who presented with bilateral daily headaches that closely resembled migraine, highlighting the potential for diagnostic confusion between the two conditions.12
The decision to pursue GCA was based on high clinical concern, and the patient’s observation of temple tenderness and prominence of his temporal arteries. This diagnosis could have easily been overlooked.
Treatment
Initial management focused primarily on post-concussive headaches with migrainous features. He was prescribed Verapamil 180 mg daily for headache prevention and rizatriptan 10 mg for acute headache management. Despite some improvement in headache intensity, the frequency of headaches remained unchanged, prompting an increase in verapamil dosage to 240 mg. After a second moderate head injury in December 2022, his symptoms worsened, and verapamil was replaced by propranolol 120 mg daily. However, he returned to verapamil due to worsening headaches with propranolol. In March 2023, the patient’s treatment was adjusted by adding venlafaxine which was titrated up to 150 mg daily in 3 weeks due to worsening symptoms. His headaches remained daily, and he experienced dizziness and concentration issues. He developed stabbing pain radiating from his right temple to his neck and jaw.
When he was found to have temporal tenderness and protruding temporal arteries, he was started on prednisone 40 mg daily for a short 4 day-course, which provided partial relief of his temple pain. His prednisone course was prolonged after GCA diagnosis for nine months which provided further improvement with the temporal headaches. However, his holocephalic headaches and light sensitivity continued, so additional migraine-specific treatment was initiated due to the headache with migraine features. Eptinezumab infusions were initiated August 2023. By February 2024, his headache severity and frequency decreased >50%, and his temporal pain improved with mild residual temporal tenderness. As of early 2025, the patient remains stable but continues to experience mild ongoing symptoms of headache, photophobia, and phonophobia. He has occasional vertigo, dizziness, and cognitive difficulties, with improved severity.
Outcome And Follow-Up
The patient continues to follow up with his primary care provider and headache specialist regularly. The most recent follow-up data was obtained in early 2025. He is scheduled to have ongoing follow-up visits every three to six months at the Duke Headache Clinic, depending on the progression of his symptoms and treatment adjustments. The patient is regularly monitored for signs of disease progression and complications. For GCA, he undergoes routine assessments, including physical examination (that includes temple palpation) and testing ESR and CRP levels for any clinical change (despite lack of elevation of ESR and CRP in the past).
Discussion
The brain injury from concussion involves metabolic, physiological and microstructural disturbances. Concussion and mild TBI has been used interchangeably in both in the medical literature and in clinical care of TBI patient population.13 Mild traumatic brain injury (mTBI), as defined by the Mild TBI Committee of the American Congress of Rehabilitation Medicine and later refined by the World Health Organization (WHO), is characterized by a Glasgow Coma Scale (GCS) score between 13 and 15, measured 30 minutes after the injury. Diagnosis requires the presence of at least one of the following symptoms: loss of consciousness lasting less than 30 minutes, post-traumatic amnesia lasting less than 24 hours, a temporary alteration in mental state at the time of the injury (such as confusion or disorientation), or a brief neurological abnormality (such as vision or balance disturbances).14 Our patient was determined to have a mild-TBI. Non-sports related concussions may result in more prolonged recovery or persistent symptoms.15
Post-concussive syndrome (PCS) refers to a group of physical, cognitive, behavioral, and emotional symptoms that appear after a traumatic brain injury (TBI). 15 These may include headaches, fatigue, vision problems, balance issues, confusion, dizziness, insomnia, neuropsychiatric symptoms, and trouble concentrating. Multiple concussions, older age, female, depression and anxiety, and chronic pain have an increased risk of developing prolonged post-concussive symptoms.16 Our patient had risk factors that could prolong recovery, including multiple concussions, age, anxiety, and chronic pain.
Computed tomography (CT) of the head is recommended for urgent evaluation. The Canadian CT Head Rule, a highly sensitive clinical decision tool was designed to standardize and enhance the emergency management of patients with minor head injury.17 Steyerberg et al. 2019, recommends that persistent neurological deficit not explained by CT is the primary indication for Brain MRI. MRI is more sensitive for detecting contusions, small hemorrhages, and axonal injuries. Approximately 30% of hospitalized TBI patients with normal head CT have abnormalities on Brain MRI.18 Functional MRI, diffusion tensor imaging (DTI), and serum biomarker are currently used for research purposes. The ability of these tools to detect structural damage and evaluate injury severity enables healthcare providers to develop appropriate treatment plans.19
There are no reported cases in literature reporting GCA developing in post-concussion syndrome patients with new onset of headaches. GCA’s pathogenesis involves an immune response that recruits inflammatory cells, leading to vascular damage and systemic inflammation.20 GCA manifests with headaches in 66% 21 of cases, jaw claudication in 50%,22 visual disturbances in 20-30%,23 and fever in 40%.24 The reason for immediate treatment is that the vasculitis ischemic complications, including anterior ischemic optic neuropathy, potentially leading to permanent vision loss.25 Between 40% and 60% of GCA patients have concomitant polymyalgia rheumatica (PMR),26 which typically presents with shoulder and pelvic girdle pain, morning stiffness, and, in some cases, systemic symptoms like fatigue, fever, and weight loss, likely driven by IL-6 signaling.27 The exact cause of GCA remains unknown, but genetic factors, such as polymorphisms in the major histocompatibility complex region, are believed to play a role.28 The incidence of GCA is higher in Scandinavian populations likely due to genetic susceptibility or immune-mediated hypersensitivity.29, 30 The diagnosis of GCA depends on patients’ clinical symptoms, lab results, ophthalmology evaluation, imaging, and/or temporal artery biopsy.
Laboratory tests, including ESR and CRP, can support diagnosis but are not specific, as normal levels may still occur in some cases.31 Several factors can lead to falsely normal ESR, including medications such as NSAIDs and statins.32 Clinicians should be aware that the use of NSAIDs or statins is linked to lower ESR levels in patients with GCA, which may reduce the sensitivity and specificity of this test for diagnosing GCA; in such cases, CRP may serve as a more reliable marker for evaluation. Steroids, including parenteral steroid administration, can lower both ESR and CRP. 33
In a retrospective, population-based cohort study, 8.2% of patients with GCA experienced permanent vision loss, primarily due to arteritic ischemic optic neuropathy (AION).34 Key diagnostic clues include pallid optic nerve head edema on slit lamp examination, delayed optic nerve head and choroidal filling on fluorescein angiography, and red flags such as bilateral or rapidly sequential vision loss and recurrence in the same eye.35 Our patient had no ophthalmologic complications.
In addition to laboratory results, color Doppler ultrasound (CDUS) of the temporal artery is now considered first-line for diagnosis because it has a higher sensitivity than temporal artery biopsy and is of lower cost and less invasive. The hallmark “halo sign,” visible on ultrasound, indicates inflammation of the vessel walls. Bilateral temporal “halo sign” is highly specific for GCA.36 In the 2016 TABUL study37 (a multi-center, prospective study for GCA), ultrasound had sensitivity of 54% and specificity of 81%, while temporal artery biopsy had sensitivity of 39% and specificity of 100%. Temporal artery biopsy remains the gold standard for diagnosis. The lower sensitivity of biopsy is likely due to skip lesions and prior corticosteroid treatment. Unexpectedly, our patient’s ultrasound was negative but biopsy was positive for GCA. Temporal artery abnormalities, including beading (irregular contour), prominence, tenderness, and an absent pulse, are reported to significantly increase the likelihood ratio for a positive temporal artery biopsy (TAB).38 Our patient had prominent temporal arteries on examination, giving him an increased likelihood of positive biopsy.
Ultrasound halo size abnormalities reduced after four or more days of high-dose glucocorticoid treatment. The percentage of positive biopsy results dropped significantly when patients began glucocorticoid treatment within 3 days.39 Our patient had the ultrasound performed approximately 20 days before starting the steroids, so the absence of the halo sign cannot be attributed to the steroids.
The first-line treatment for GCA includes high-dose corticosteroids, which help reduce inflammation to prevent severe conditions like vision loss and stroke.40 The American College of Rheumatology (ACR) recommends starting with 40 to 60 mg of prednisone daily, tapering once symptoms improve, which needs to be done gradually due to risk of developing adrenal insufficiency.41 In cases with persistent or worsening symptoms or contraindications to prednisone, like uncontrolled diabetes, tocilizumab (an IL-6 inhibitor) or methotrexate may be used.42 Two RCTs involving 281 participants found that tocilizumab significantly improved sustained remission and reduced relapse time in giant cell arteritis, with the weekly dose showing the best results.43 For patients with significant vascular involvement, low-dose aspirin is also recommended.44
Early diagnosis and immediate treatment are crucial in preventing irreversible damage and complications such as strokes, especially in elderly patients due to the presence of other associated vascular risk factors.45 Most strokes secondary to GCA are ischemic in nature, with only a few isolated cases reporting subarachnoid hemorrhage as a complication of GCA.46 GCA-related strokes typically occur in the vertebrobasilar (VB) territory and are more common in patients who experience ophthalmic ischemic symptoms.47 In a case series, the authors discuss three patients with biopsy-proven GCA who experienced significant auditory and vestibular symptoms.48 Two of these patients died due to GCA affecting the vertebral arteries, with postmortem findings reported, emphasizing the need for a high index of suspicion for GCA in elderly patients with acute audiovestibular symptoms like deafness, tinnitus, vertigo and loss of balance,49 as they may signal brainstem infarction. Carotid and vertebral artery dissections have been documented, as well.50
Conclusion
In conclusion, both PCS and GCA require careful evaluation and management due to their potential long-term impacts on patients’ health. PCS, resulting from mild traumatic brain injury, manifests in a range of persistent symptoms, with treatment focusing on symptom relief and rehabilitation. GCA, a serious inflammatory condition affecting large arteries, demands prompt diagnosis and intervention to prevent severe complications. Physical examination and high clinical suspicion are needed to appropriately diagnose patients with comorbid conditions. Continued research into the mechanisms and management of these conditions will further enhance clinical care and patient quality of life.
References
- Dejaco C, Brouwer E, Mason JC, Buttgereit F, Matteson EL, Dasgupta B. Giant cell arteritis and polymyalgia rheumatica: current challenges and opportunities. Nat Rev Rheumatol. Oct 2017;13(10):578-592. PubMed PMID: 28905861. doi:10.1038/nrrheum.2017.142
- Headache Classification Committee of the International Headache S. The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia. Jul 2013;33(9):629-808. PubMed PMID: 23771276. doi:10.1177/0333102413485658
- Stamatis P, Turesson C, Mohammad AJ. Temporal artery biopsy in giant cell arteritis: clinical perspectives and histological patterns. Front Med (Lausanne). 2024;11:1453462. PubMed PMID: 39386746; PubMed Central PMCID: PMCPMC11461189. doi:10.3389/fmed.2024.1453462
- Ishii R, Schwedt TJ, Trivedi M, et al. Mild traumatic brain injury affects the features of migraine. J Headache Pain. Jul 22 2021;22(1):80. PubMed PMID: 34294026; PubMed Central PMCID: PMCPMC8296591. doi:10.1186/s10194-021-01291-x
- Anderson GC, John MT, Ohrbach R, et al. Influence of headache frequency on clinical signs and symptoms of TMD in subjects with temple headache and TMD pain. Pain. Apr 2011;152(4):765-771. PubMed PMID: 21196079; PubMed Central PMCID: PMCPMC3065515. doi:10.1016/j.pain.2010.11.007
- Ashkenazi A, Sholtzow M, Shaw JW, Burstein R, Young WB. Identifying cutaneous allodynia in chronic migraine using a practical clinical method. Cephalalgia. Feb 2007;27(2):111-7. PubMed PMID: 17257230; PubMed Central PMCID: PMCPMC2664546. doi:10.1111/j.1468-2982.2006.01255.x
- Markus TE, Zeharia A, Cohen YH, Konen O. Persistent Headache and Cephalic Allodynia Attributed to Head Trauma in Children and Adolescents. J Child Neurol. Sep 2016;31(10):1213-9. PubMed PMID: 27221373. doi:10.1177/0883073816650036
- Pijpers JA, Kies DA, van Zwet EW, de Boer I, Terwindt GM. Cutaneous allodynia as predictor for treatment response in chronic migraine: a cohort study. J Headache Pain. Aug 30 2023;24(1):118. PubMed PMID: 37644420; PubMed Central PMCID: PMCPMC10466691. doi:10.1186/s10194-023-01651-9
- Laldinpuii J, Sanchetee P, Borah AL, Ghose M, Borah NC. Giant cell arteritis (temporal arteritis): A report of four cases from north east India. Ann Indian Acad Neurol. Jul 2008;11(3):185-9. PubMed PMID: 19893667; PubMed Central PMCID: PMCPMC2771971. doi:10.4103/0972-2327.42940
- Tripakornkusol V, Sinsopa N, Khamsai S, Sawanyawisuth K. Phenotypes of headache in patients with obstructive sleep apnea. Sci Rep. Feb 8 2025;15(1):4806. PubMed PMID: 39922920; PubMed Central PMCID: PMCPMC11807175. doi:10.1038/s41598-025-89538-4
- Ashina S, Mitsikostas DD, Lee MJ, et al. Tension-type headache. Nat Rev Dis Primers. Mar 25 2021;7(1):24. PubMed PMID: 33767185. doi:10.1038/s41572-021-00257-2
- Devi S, Dash A, Purkait S, Sahoo B. Giant Cell Arteritis Masquerading As Migraine: A Case Report. Cureus. Aug 2023;15(8):e44107. PubMed PMID: 37750130; PubMed Central PMCID: PMCPMC10518159. doi:10.7759/cureus.44107
- Sussman ES, Pendharkar AV, Ho AL, Ghajar J. Mild traumatic brain injury and concussion: terminology and classification. Handb Clin Neurol. 2018;158:21-24. PubMed PMID: 30482349. doi:10.1016/B978-0-444-63954-7.00003-3
- Lefevre-Dognin C, Cogne M, Perdrieau V, Granger A, Heslot C, Azouvi P. Definition and epidemiology of mild traumatic brain injury. Neurochirurgie. May 2021;67(3):218-221. PubMed PMID: 32387427. doi:10.1016/j.neuchi.2020.02.002
- Gaudet LA, Eliyahu L, Beach J, et al. Workers’ recovery from concussions presenting to the emergency department. Occup Med (Lond). Oct 1 2019;69(6):419-427. PubMed PMID: 31340019; PubMed Central PMCID: PMCPMC6769822. doi:10.1093/occmed/kqz089
- Bazarian JJ, Wong T, Harris M, Leahey N, Mookerjee S, Dombovy M. Epidemiology and predictors of post-concussive syndrome after minor head injury in an emergency population. Brain Inj. Mar 1999;13(3):173-89. PubMed PMID: 10081599. doi:10.1080/026990599121692
- Stiell IG, Wells GA, Vandemheen K, et al. The Canadian CT Head Rule for patients with minor head injury. Lancet. May 5 2001;357(9266):1391-6. PubMed PMID: 11356436. doi:10.1016/s0140-6736(00)04561-x
- Steyerberg EW, Wiegers E, Sewalt C, et al. Case-mix, care pathways, and outcomes in patients with traumatic brain injury in CENTER-TBI: a European prospective, multicentre, longitudinal, cohort study. Lancet Neurol. Oct 2019;18(10):923-934. PubMed PMID: 31526754. doi:10.1016/S1474-4422(19)30232-7
- Leddy J, Baker JG, Haider MN, Hinds A, Willer B. A Physiological Approach to Prolonged Recovery From Sport-Related Concussion. J Athl Train. Mar 2017;52(3):299-308. PubMed PMID: 28387557; PubMed Central PMCID: PMCPMC5384826. doi:10.4085/1062-6050-51.11.08
- Deng J, Younge BR, Olshen RA, Goronzy JJ, Weyand CM. Th17 and Th1 T-cell responses in giant cell arteritis. Circulation. Feb 23 2010;121(7):906-15. PubMed PMID: 20142449; PubMed Central PMCID: PMCPMC2837465. doi:10.1161/CIRCULATIONAHA.109.872903
- Dejaco C, Duftner C, Buttgereit F, Matteson EL, Dasgupta B. The spectrum of giant cell arteritis and polymyalgia rheumatica: revisiting the concept of the disease. Rheumatology (Oxford). Apr 1 2017;56(4):506-515. PubMed PMID: 27481272. doi:10.1093/rheumatology/kew273
- Goyal N, Basnet A, Donenfeld TT, et al. An Unusual Case of Giant Cell Arteritis. Cureus. Jul 2022;14(7):e26483. PubMed PMID: 35919218; PubMed Central PMCID: PMCPMC9339147. doi:10.7759/cureus.26483
- Smetana GW, Shmerling RH. Does this patient have temporal arteritis? JAMA. Jan 2 2002;287(1):92-101. PubMed PMID: 11754714. doi:10.1001/jama.287.1.92
- Gonzalez-Gay MA, Garcia-Porrua C, Amor-Dorado JC, Llorca J. Fever in biopsy-proven giant cell arteritis: clinical implications in a defined population. Arthritis Rheum. Aug 15 2004;51(4):652-5. PubMed PMID: 15334440. doi:10.1002/art.20523
- Gabriel SE, O’Fallon WM, Achkar AA, Lie JT, Hunder GG. The use of clinical characteristics to predict the results of temporal artery biopsy among patients with suspected giant cell arteritis. J Rheumatol. Jan 1995;22(1):93-6. PubMed PMID: 7699690.
- Cantini F, Niccoli L, Storri L, et al. Are polymyalgia rheumatica and giant cell arteritis the same disease? Semin Arthritis Rheum. Apr 2004;33(5):294-301. PubMed PMID: 15079760. doi:10.1016/j.semarthrit.2003.09.008
- Lundberg IE, Sharma A, Turesson C, Mohammad AJ. An update on polymyalgia rheumatica. J Intern Med. Nov 2022;292(5):717-732. PubMed PMID: 35612524; PubMed Central PMCID: PMCPMC9796644. doi:10.1111/joim.13525
- Carmona FD, Mackie SL, Martin JE, et al. A large-scale genetic analysis reveals a strong contribution of the HLA class II region to giant cell arteritis susceptibility. Am J Hum Genet. Apr 2 2015;96(4):565-80. PubMed PMID: 25817017; PubMed Central PMCID: PMCPMC4385191. doi:10.1016/j.ajhg.2015.02.009
- Pucelj NP, Hocevar A, Jese R, et al. The incidence of giant cell arteritis in Slovenia. Clin Rheumatol. Feb 2019;38(2):285-290. PubMed PMID: 30062445. doi:10.1007/s10067-018-4236-6
- Lee JL, Naguwa SM, Cheema GS, Gershwin ME. The geo-epidemiology of temporal (giant cell) arteritis. Clin Rev Allergy Immunol. Oct 2008;35(1-2):88-95. PubMed PMID: 18286386. doi:10.1007/s12016-008-8075-0
- Kermani TA, Schmidt J, Crowson CS, et al. Utility of erythrocyte sedimentation rate and C-reactive protein for the diagnosis of giant cell arteritis. Semin Arthritis Rheum. Jun 2012;41(6):866-71. PubMed PMID: 22119103; PubMed Central PMCID: PMCPMC3307891. doi:10.1016/j.semarthrit.2011.10.005
- Hegg R, Lee AG, Tagg NT, Zimmerman MB. Statin or nonsteroidal anti-inflammatory drug use is associated with lower erythrocyte sedimentation rate in patients with giant cell arteritis. J Neuroophthalmol. Jun 2011;31(2):135-8. PubMed PMID: 21358421. doi:10.1097/WNO.0b013e31820c4421
- Taylor HG, Fowler PD, David MJ, Dawes PT. Intra-articular steroids: confounder of clinical trials. Clin Rheumatol. Mar 1991;10(1):38-42. PubMed PMID: 2065506. doi:10.1007/BF02208031
- Chen JJ, Leavitt JA, Fang C, Crowson CS, Matteson EL, Warrington KJ. Evaluating the Incidence of Arteritic Ischemic Optic Neuropathy and Other Causes of Vision Loss from Giant Cell Arteritis. Ophthalmology. Sep 2016;123(9):1999-2003. PubMed PMID: 27297405; PubMed Central PMCID: PMCPMC4995137. doi:10.1016/j.ophtha.2016.05.008
- Yang Wu K EF. NAION: Diagnosis and Management. Accessed May 2025, 2025. https://www.aao.org/eyenet/article/naion-diagnosis-and-management
- Mackie SL, Dejaco C, Appenzeller S, et al. British Society for Rheumatology guideline on diagnosis and treatment of giant cell arteritis. Rheumatology (Oxford). Mar 1 2020;59(3):e1-e23. PubMed PMID: 31970405. doi:10.1093/rheumatology/kez672
- Luqmani R, Lee E, Singh S, et al. The Role of Ultrasound Compared to Biopsy of Temporal Arteries in the Diagnosis and Treatment of Giant Cell Arteritis (TABUL): a diagnostic accuracy and cost-effectiveness study. Health Technol Assess. Nov 2016;20(90):1-238. PubMed PMID: 27925577; PubMed Central PMCID: PMCPMC5165283. doi:10.3310/hta20900
- Lyons HS, Quick V, Sinclair AJ, Nagaraju S, Mollan SP. A new era for giant cell arteritis. Eye (Lond). Jun 2020;34(6):1013-1026. PubMed PMID: 31582795; PubMed Central PMCID: PMCPMC7253415. doi:10.1038/s41433-019-0608-7
- Koster MJ, Yeruva K, Crowson CS, Muratore F, Labarca C, Warrington KJ. Efficacy of Methotrexate in Real-world Management of Giant Cell Arteritis: A Case-control Study. J Rheumatol. May 2019;46(5):501-508. PubMed PMID: 30647171. doi:10.3899/jrheum.180429
- Chung SA, Langford CA, Maz M, et al. 2021 American College of Rheumatology/Vasculitis Foundation Guideline for the Management of Antineutrophil Cytoplasmic Antibody-Associated Vasculitis. Arthritis Rheumatol. Aug 2021;73(8):1366-1383. PubMed PMID: 34235894; PubMed Central PMCID: PMCPMC12327957. doi:10.1002/art.41773
- Hellmich B, Agueda A, Monti S, et al. 2018 Update of the EULAR recommendations for the management of large vessel vasculitis. Ann Rheum Dis. Jan 2020;79(1):19-30. PubMed PMID: 31270110. doi:10.1136/annrheumdis-2019-215672
- Grazzini S, Conticini E, Falsetti P, et al. Tocilizumab Vs Methotrexate in a Cohort of Patients Affected by Active GCA: A Comparative Clinical and Ultrasonographic Study. Biologics. 2023;17:151-160. PubMed PMID: 38059132; PubMed Central PMCID: PMCPMC10697083. doi:10.2147/BTT.S431818
- Antonio AA, Santos RN, Abariga SA. Tocilizumab for giant cell arteritis. Cochrane Database Syst Rev. May 13 2022;5(5):CD013484. PubMed PMID: 35560150; PubMed Central PMCID: PMCPMC9105486. doi:10.1002/14651858.CD013484.pub3
- Nesher G, Berkun Y, Mates M, Baras M, Rubinow A, Sonnenblick M. Low-dose aspirin and prevention of cranial ischemic complications in giant cell arteritis. Arthritis Rheum. Apr 2004;50(4):1332-7. PubMed PMID: 15077317. doi:10.1002/art.20171
- Samson M, Jacquin A, Audia S, et al. Stroke associated with giant cell arteritis: a population-based study. J Neurol Neurosurg Psychiatry. Feb 2015;86(2):216-21. PubMed PMID: 24780954. doi:10.1136/jnnp-2014-307614
- Takahashi I, Takamura H, Gotoh S, Sasaki H, Ishikawa T. Giant cell arteritis with subarachnoid haemorrhage due to the rupture of inflammatory aneurysm of the posterior inferior cerebellar artery. Acta Neurochir (Wien). 1996;138(7):893-4. PubMed PMID: 8869722. doi:10.1007/BF01411272
- Bajko Z, Balasa R, Maier S, et al. Stroke secondary to giant-cell arteritis: A literature review. Exp Ther Med. Aug 2021;22(2):876. PubMed PMID: 34194554; PubMed Central PMCID: PMCPMC8237390. doi:10.3892/etm.2021.10308
- McKennan KX, Nielsen SL, Watson C, Wiesner K. Meniere’s syndrome: an atypical presentation of giant cell arteritis (temporal arteritis). Laryngoscope. Oct 1993;103(10):1103-7. PubMed PMID: 8412445. doi:10.1288/00005537-199310000-00005
- Saravanan V, Pugmire S, Smith M, Kelly C. Patient-reported involvement of the eighth cranial nerve in giant cell arteritis. Clin Rheumatol. Dec 2019;38(12):3655-3660. PubMed PMID: 31420810. doi:10.1007/s10067-019-04747-3
- Bonnan M, Debeugny S. Giant-cell arteritis related strokes: scoping review of mechanisms and rethinking treatment strategy? Front Neurol. 2023;14:1305093. PubMed PMID: 38130834; PubMed Central PMCID: PMCPMC10733536. doi:10.3389/fneur.2023.1305093
Acknowledgement
Jacob Curran, Technical Director of Neurovascular Sonography, Duke University Medical Center
Consent
Consent was obtained from the patient for the publication of this case report, including all relevant clinical information and imaging.
Declarations/Disclosures
Funding/Conflicts of interest: In compliance with the ICMJE uniform disclosure form, author declares the following:
Payment/services info: Author has declared that no financial support was received from any organization for the submitted work.
Financial relationships: Author have declared that they have no financial relationships at present or within the previous three years with any organizations that might have an interest in the submitted work.
Other relationships: Author have declared that there are no other relationships or activities that could appear to have influenced the submitted work.
